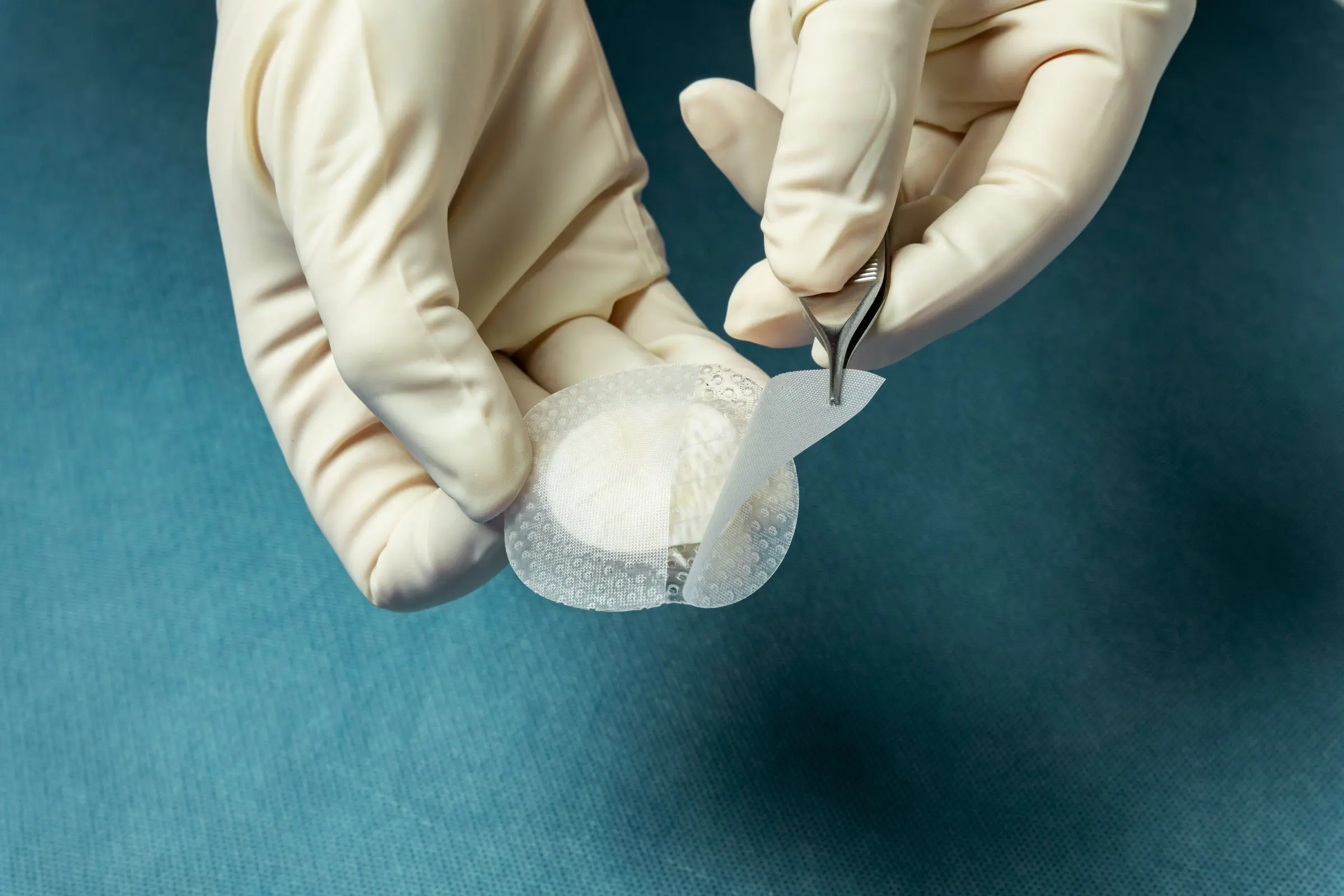
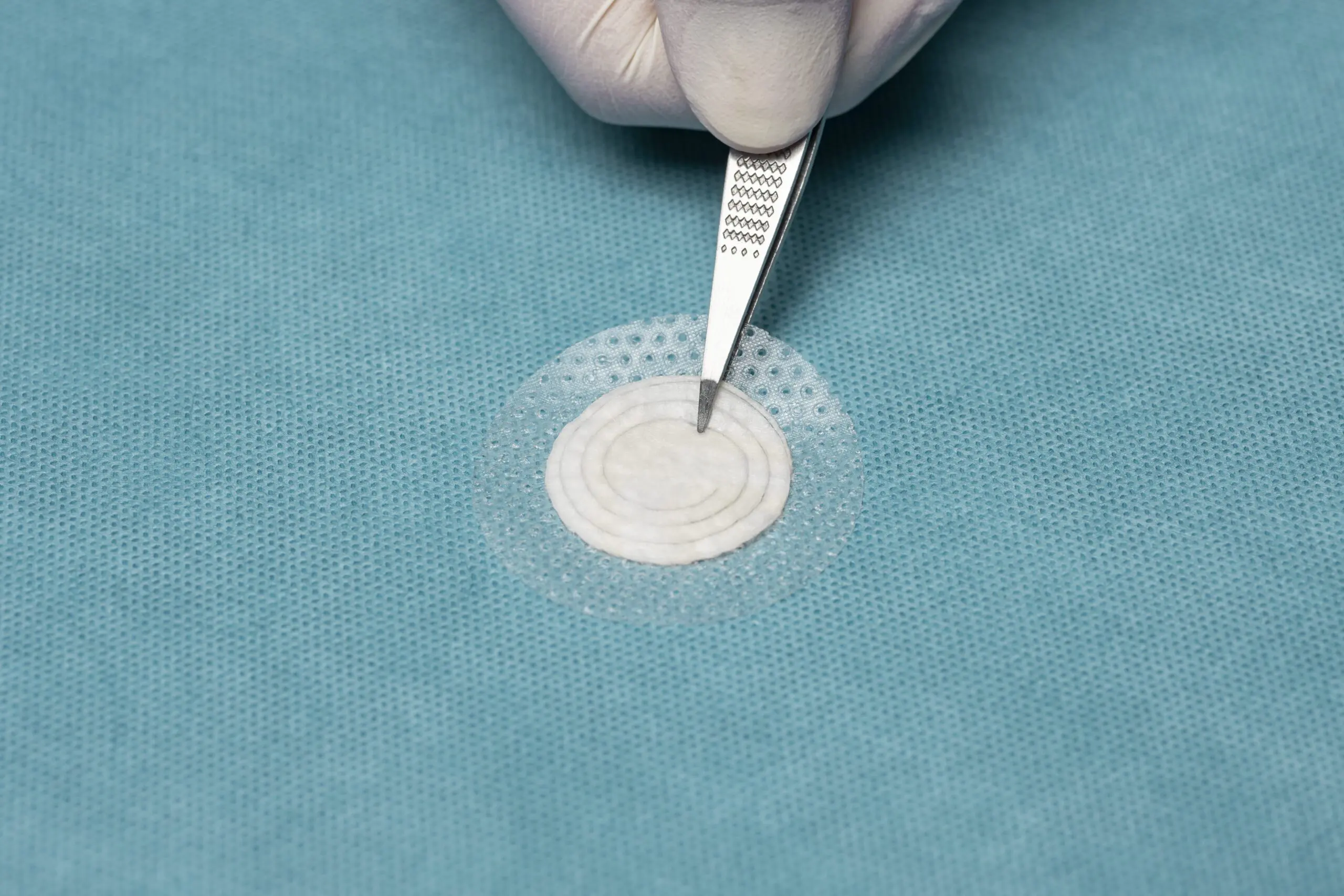
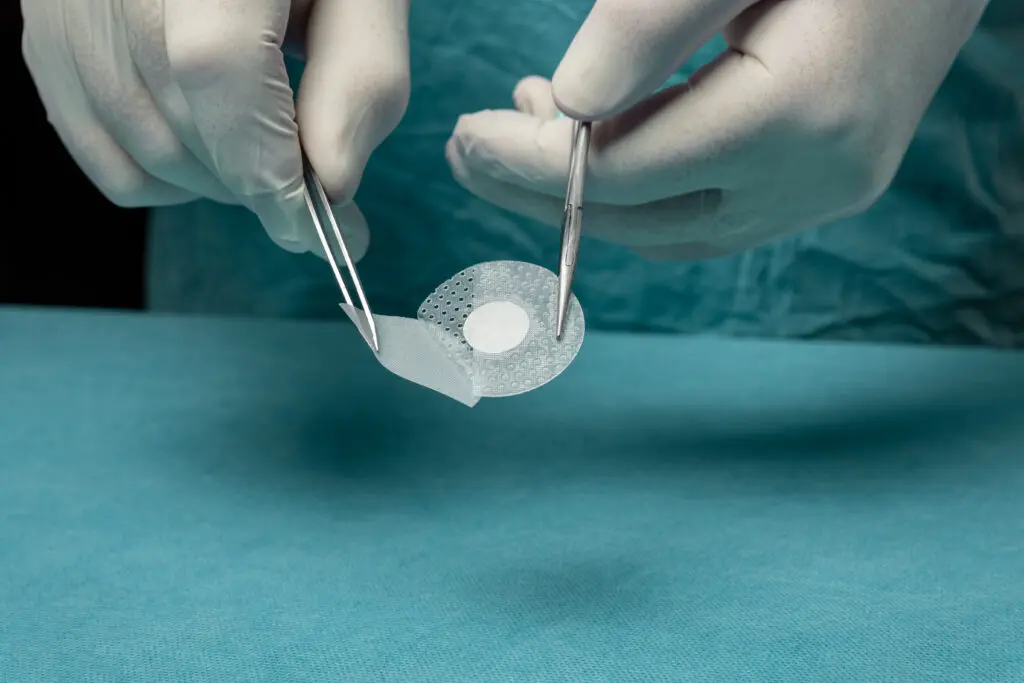
The Four Hallmarks of Kerecis Tissue Regeneration
Kerecis' patented fish skin is gently processed, preserving its homologous structure to human skin. These four hallmarks are recognized as optimal traits for a tissue replacement product.
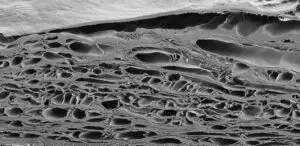
Intact Molecular Organization
Fish skin is homologous to human skin, with a comparable pore structure that facilitates rapid cellular ingrowth. The gentle EnviroIntact™ process preserves the native extracellular matrix architecture — including the natural arrangement of collagen fibers, elastin, and glycosaminoglycans — creating an ideal scaffold for host cell infiltration and tissue formation.

Preserved Molecular Content
Because there is no need for harsh antiviral processing, the chemical complexity of the fish skin is preserved — including omega-3 polyunsaturated fatty acids (DHA and EPA) that activate specialized pro-resolving mediator (SPM) pathways. These reduce IL-1β production from macrophages, resolving inflammation while promoting healing. Fish skin has been shown to facilitate 20x more cell ingrowth than amniotic tissue.[DOI]

Three-Dimensional Structure
The intact structure of the fish skin provides a framework to support tissue regeneration. The natural porosity enables rapid ingrowth of fibroblasts (connective tissue) and blood vessels. When grafted onto a wound, human cells migrate into the fish skin pores and start forming connective tissue and facilitating neovascularization. The body incorporates the fish skin within 5–7 days.

Natural Mechanical Properties
Kerecis fish skin is naturally strong, handles like skin, and is easy to suture or staple. It provides an immediate natural microbial barrier that protects against environmental insults while the wound transitions from the inflammatory to proliferative phase. The product resists shearing forces, allowing early physiotherapy — particularly important in burn management.