Non-Opioid Pain Management
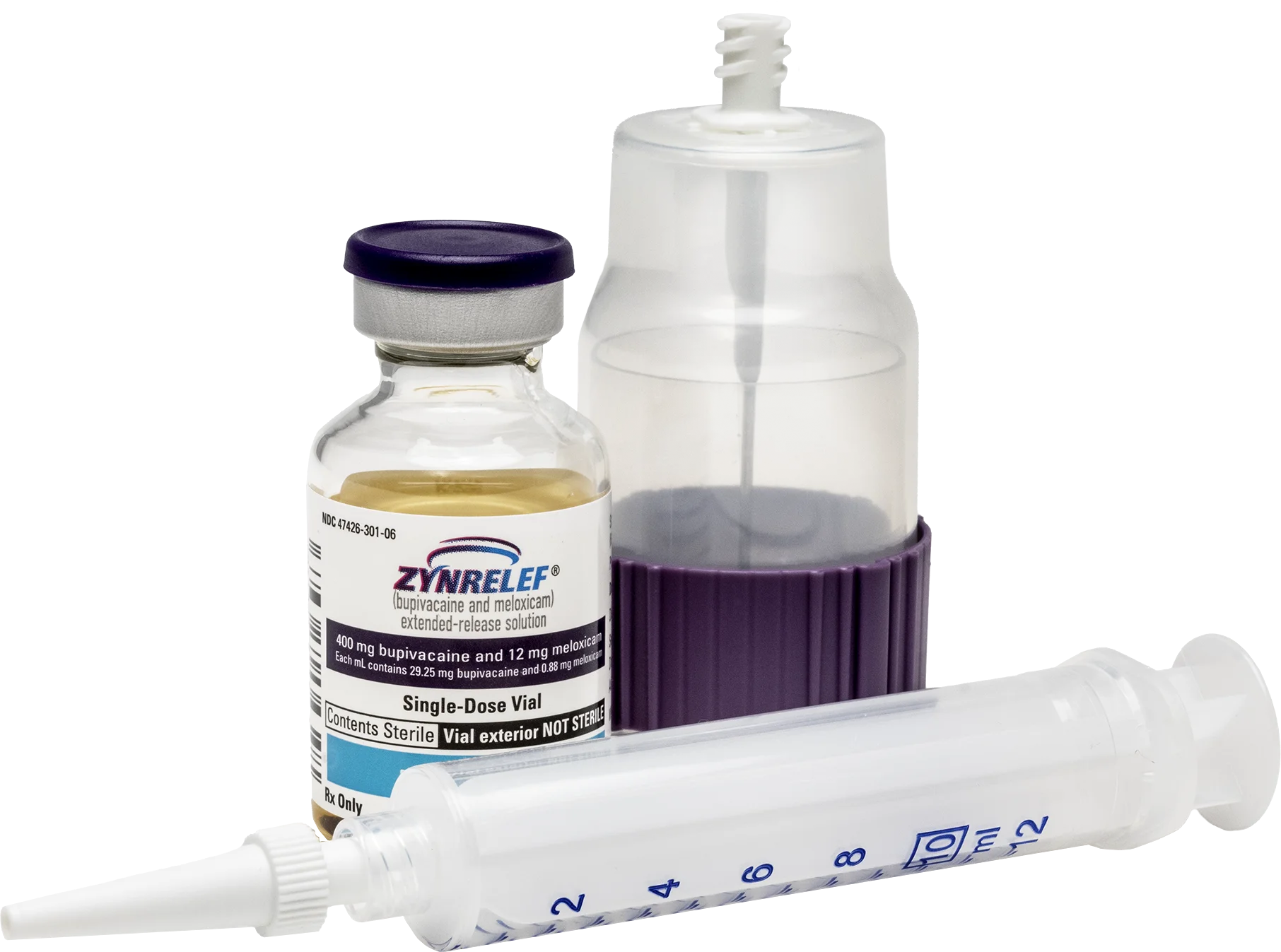
ZYNRELEF
The first and only dual-acting extended-release local anesthetic — combining bupivacaine with low-dose meloxicam for up to 72 hours of postoperative pain relief.
Applied directly to the surgical site via needle-free applicator. No injection. No reconstitution. One application.
Request Product Information View Clinical Evidence